14-78 The wall of a house is made of a 20-cm thick brick. The amount of moisture flowing through the wall in 24-h is to be
determined.
Assumptions 1 Steady operating conditions exist. 2 Mass transfer through the wall is one-dimensional. 3 The vapor
permeability of the wall is constant.
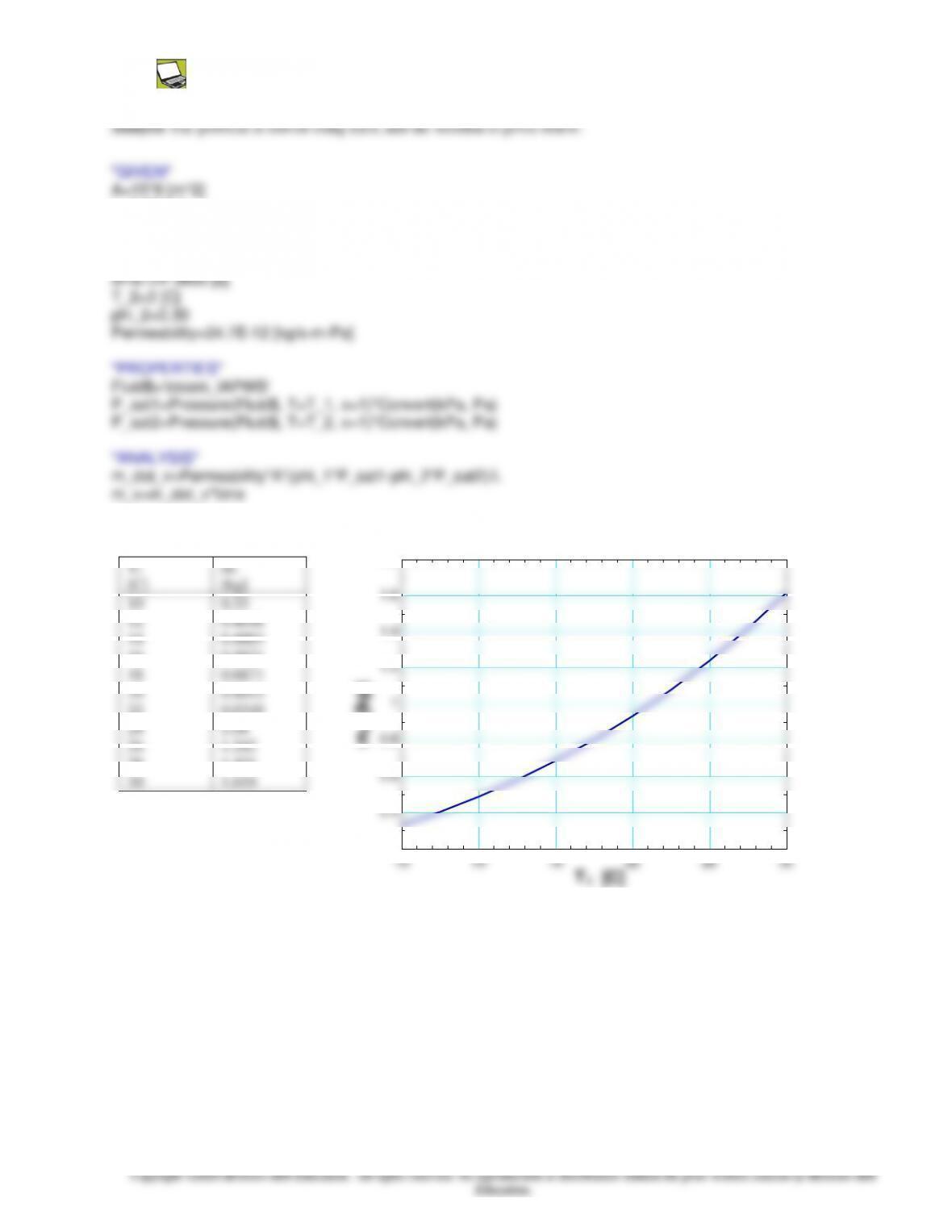
Properties The permeance of 100 mm thick wall is 4610-12
The mass flow rate of water vapor through a plain layer of
thickness L and normal area A is given as (Eq. 14-31)
L
PP
A
L
PP
Am vv
v
sat,221,sat1,21,
−
=
−
=PP
where P is the vapor permeability, is the relative humidity and Psat is the
saturation pressure of water at the specified temperature. Subscripts 1 and 2
denote the states of the air on the two sides of the roof. Substituting, the mass
flow rate of water vapor through the wall is determined to be
2813 mkg/s1088.3
m 20.0
]Pa) 3169(40.0)Pa 7384(40.0[
)kg/s.m.Pa1046(=
−
= −−
v
m
Then the total amount of moisture that flows through the roof during a 24-h period becomes
g 3.35==== −
−kg 0.00335s) 3600)(24mkg/s10(3.88 28
24,tmm vhv